Electron configurations of elements beyond hassium (element 108) have never been measured predictions. The Madelung rule gives the order: 1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s < 5f < 6d < 7p Oganesson (element 118) is a good example to show the order of the orbitals. For each atom the subshells are given first in concise form, then with all subshells written out, followed by the number of electrons per shell. However, it's easy to determine the configuration of electrons for heavier elements by making a chart. This page shows the electron configurations of the neutral gaseous atoms in their ground states. If there are more electrons than protons, the ion has a negative charge and is called an anion.Įlements are shown from atomic number 1 (hydrogen) up to 94 (plutonium). If there are more protons than electrons, an atomic ion has a positive charge and is called a cation. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons-or not.Īn ion of an atom is one in which the number of protons and electrons is not the same. 
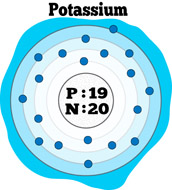
Remember, a neutral atom contains the same number of protons and electrons. The upper right side shows the number of electrons in a neutral atom. The element atomic number and name are listed in the upper left. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. 
The electron shells are shown, moving outward from the nucleus. Here are electron shell atom diagrams for the elements, ordered by increasing atomic number.įor each electron shell atom diagram, the element symbol is listed in the nucleus. For that, we have electron shell diagrams. It's easier to understand electron configuration and valence if you can actually see the electrons surrounding atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
